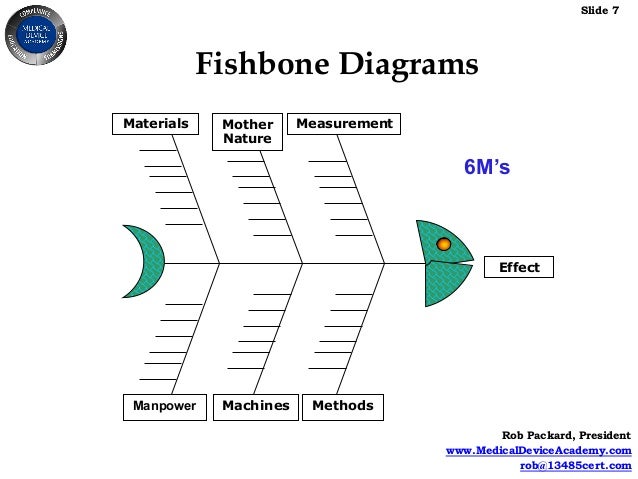

Capa processes iso#
Requirements for CAPA management as they apply to the highly regulated world of medical device development can all be found within ISO 13485:2016, the MHRA guidelines, and FDA 21 CFR Part 820.100 But what is CAPA management?ĬAPA refers to two distinct requirements for documented ‘Corrective Action’ and ‘Preventative Action’ procedures that should form part of your med tech Quality Management System. Moreover, do not hesitate to look at our EU MDR E-book and EU IVDR E-Book collecting a vast number of information on topics related to the European Medical Device Regulation 2017/745 and In-Vitro Diagnostic Regulation 2017/746.Mastering CAPA management is part of the proactive quality mindset that regulators demand, but it is also an absolute requirement for achieving commercial success in the medical device industry.
These compliance kits include different guidelines, ebooks, templates and procedures that are essentials. Recently we introduced our Compliance Kits related to EU MDR 2017/745 and post-market surveillance activities. If you have any topic for which you would like to have more information or you need template or documentation that is currently not available in our QualityMedDev Shop, do not hesitate to contact us and we will do our best to fulfil your request.

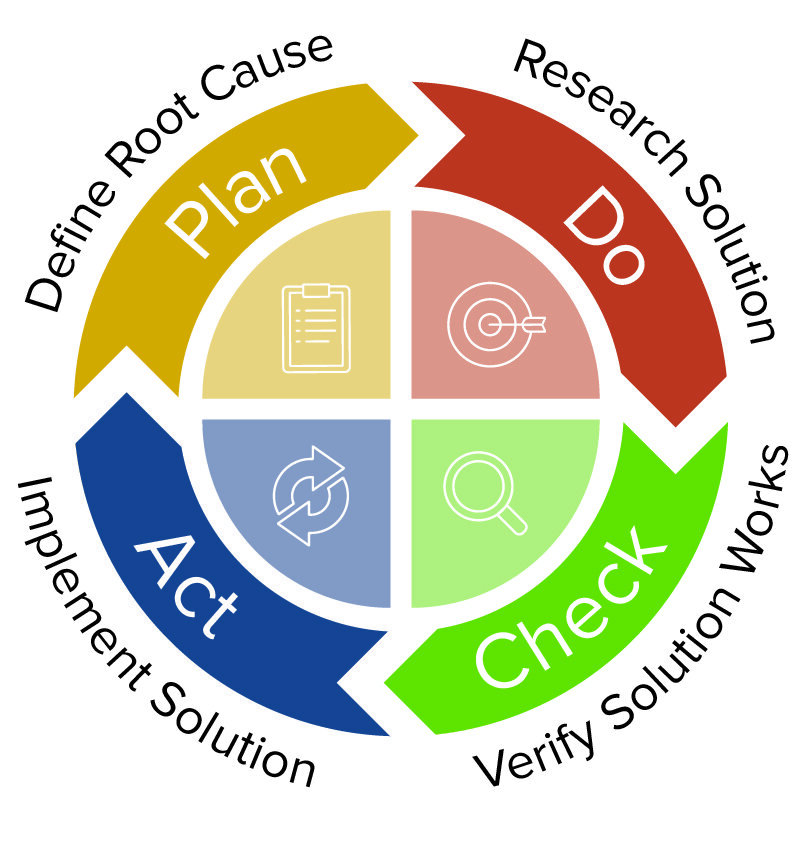
Capa processes verification#
We provide regulatory consulting services over a broad range of topics, from EU MDR & IVDR to ISO 13485, including risk management, biocompatibility, usability and software verification and validation and, in general, support in preparation of technical documentation for MDR. QualityMedDev is one of the largest online platform supporting medical device business for regulatory compliance topics. QualityMedDev is an online platform focused on Quality & Regulatory topics for medical device business Follow us on LinkedIn and Twitter to stay up to date with most important news on the Regulatory field. If, following an issue, a decision not to implement any action is taken, this should be justified using a risk-based approach, including a determination that the finished device still meets risk acceptability criteria. Corrections and corrective actions must always be commensurate with the risk associated with the nonconformity.
